Aquatic eDNA Testing
Background
Aquatic Environmental DNA, or aquatic eDNA, is a non-invasive tool used to detect the presence of fish and other aquatic lifeforms. It tests water samples for genetic material such as skin tissue, urine, and feces that has been shed by fish other animals. Aquatic eDNA is an invaluable tool for helping to understand what species of fish are present in a given lake or pond, or section of river or stream.
Aquatic eDNA can be used to determine species presence, biodiversity, and in some cases abundance. It can be used to verify the presence of specific species including rare or stressed fish, as well as for detecting the presence of nonnative invasive fish species. While aquatic eDNA can be considered proof of presence when done with the right technology and appropriate controls, it is not, however, considered proof of absence.
While targeted eDNA is proof of presence when done properly, eDNA metabarcoding is typically not considered absolute proof of presence and should be further verified by angling, seining, netting, trapping, and/or electro-fishing.
Aquatic eDNA is an effective and efficient way to cover a lot of water while being less invasive and labor intensive than angling, seining, netting, trapping, or electro-fishing.
Types of Aquatic eDNA Testing
There are two primary types of aquatic eDNA: Targeted eDNA and eDNA metabarcoding. the two technologies have very different applications and should be used accordingly.
Targeted aquatic eDNA focuses on a specific species. This is the most common form of eDNA sampling. Most targeted eDNA is done by colleges or private labs. Targeted eDNA is used when trying to confirm or determine the presence of a specific species of fish:
Rare species or subspecies where they are not known to be
Declining or possibly extirpated populations.
Used by Massachusetts NFC to determine if wild native brook trout were still present in Trout Brook in Dover, MA.
Invasive species.
eDNA metabarcoding is a relatively new technology. Rather than testing for a specific species, eDNA metabarcoding tests for a range of species. It is however less exact and less likely to be considered proof or presence.
eDNA metabarcoding typically cannot drill down as deep as targeted eDNA when it comes to species identification. For example, while targeted eDNA can differentiate between subspecies, eDNA metabarcoding often reports at the species level, or even genus.
NFC uses Jonah Ventures for eDNA metabarcoding testing.
eDNA metabarcoding is the best technology when you do not know exactly what you are looking for, or are looking for a place to start.
eDNA metabarcoding is a great tool for establishing baseline fish biodiversity in regard to Holitic Stream assessment/Restoration projects.
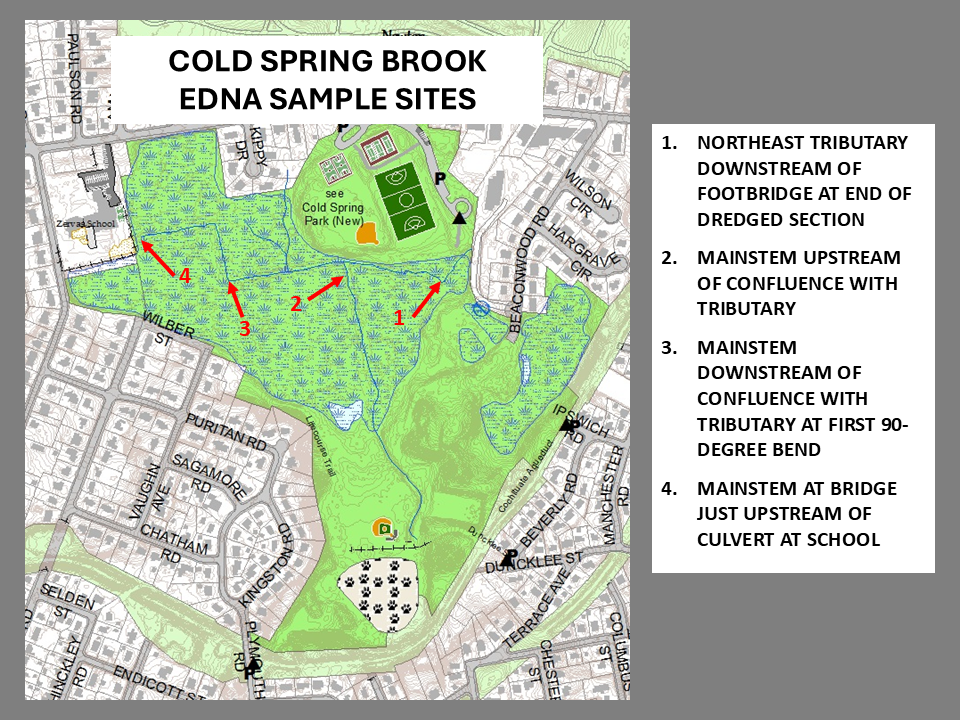
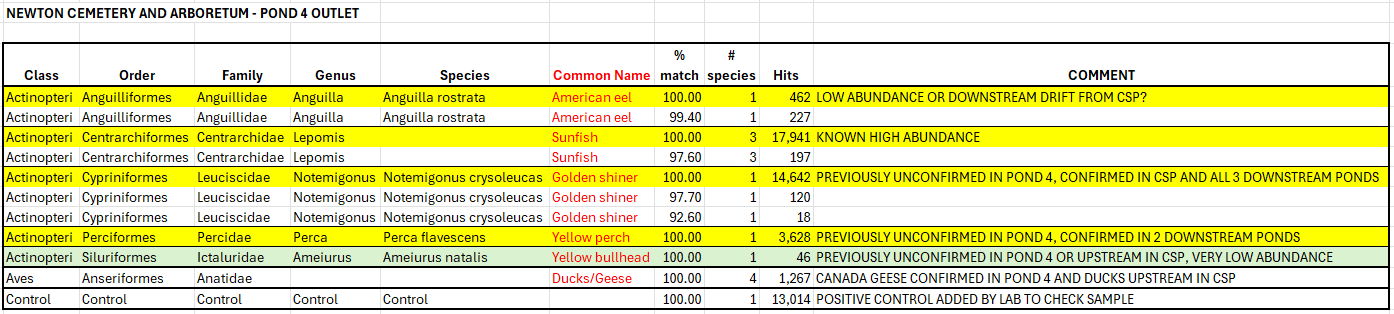
eDNA metabarcoding was used by Massachusetts NFC to determine what species of fish might be living in Cold Spring Brook in Cold Spring Park in Newton, MA.
In addition to testing positive for golden shiners which NFC had already confirmed to be present, several samples tested positive for American eel, a species no one would have thought to look for. MA NFC used this data to direct e-fishing efforts which confirmed the presence of eels.
Negative and Positive Controls
Negative and positive controls are used to strengthen the validity of the eDNA results. This is important when you are sharing the data outside of NFC or using the data to request action on the part of government agencies and others.
Negative controls are used to detect contamination that could occur in the field or at the lab. This is done by submitting a sample of distilled water or something else that does not contain any eDNA.
Negative controls are processed the same as field samples.
Negative controls cost as much as field samples.
The lab should not be made aware of negative controls.
Negative controls should show no eDNA.
NFC does not recommend negative controls for eDNA metabarcoding as most do not consider it proof of presence regardless of controls.
NFC recommends negative controls for targeted eDNA. It is said that over 90% of targeted eDNA testing includes at least one negative control.
Positive controls are used to show that the lab processes are complete and correct and can accurately detect and report the presence of eDNA. This can be done by submitting a sample that contains known eDNA in relatively high abundance from species that would not be present in the related field samples.
Positive controls are processed the same as field samples.
Positive controls cost as much as field samples.
The lab should not be made aware of positive controls.
Positive controls should only show eDNA from the introduced species.
NFC does not recommend positive controls for eDNA metabarcoding as most do not consider it proof of presence regardless of controls.
NFC recommends positive controls for targeted eDNA.
Labs sometimes introduce their own positive controls.
Sample Collection
Both targeted eDNA and eDNA metabarcoding can be tested directly from water or via a filter.
All labs provide sample collection instructions. It is critical that you follow the instructions carefully and completely.
NFC will provide chapters with laminated instruction cards for Jonah Ventures eDNA metabarcoding sample collection. See below.
Sterilize all containers when submitting water samples.
Always wear rubber gloves when handling samples.
When using water samples, you must keep the samples cool and free of contamination.
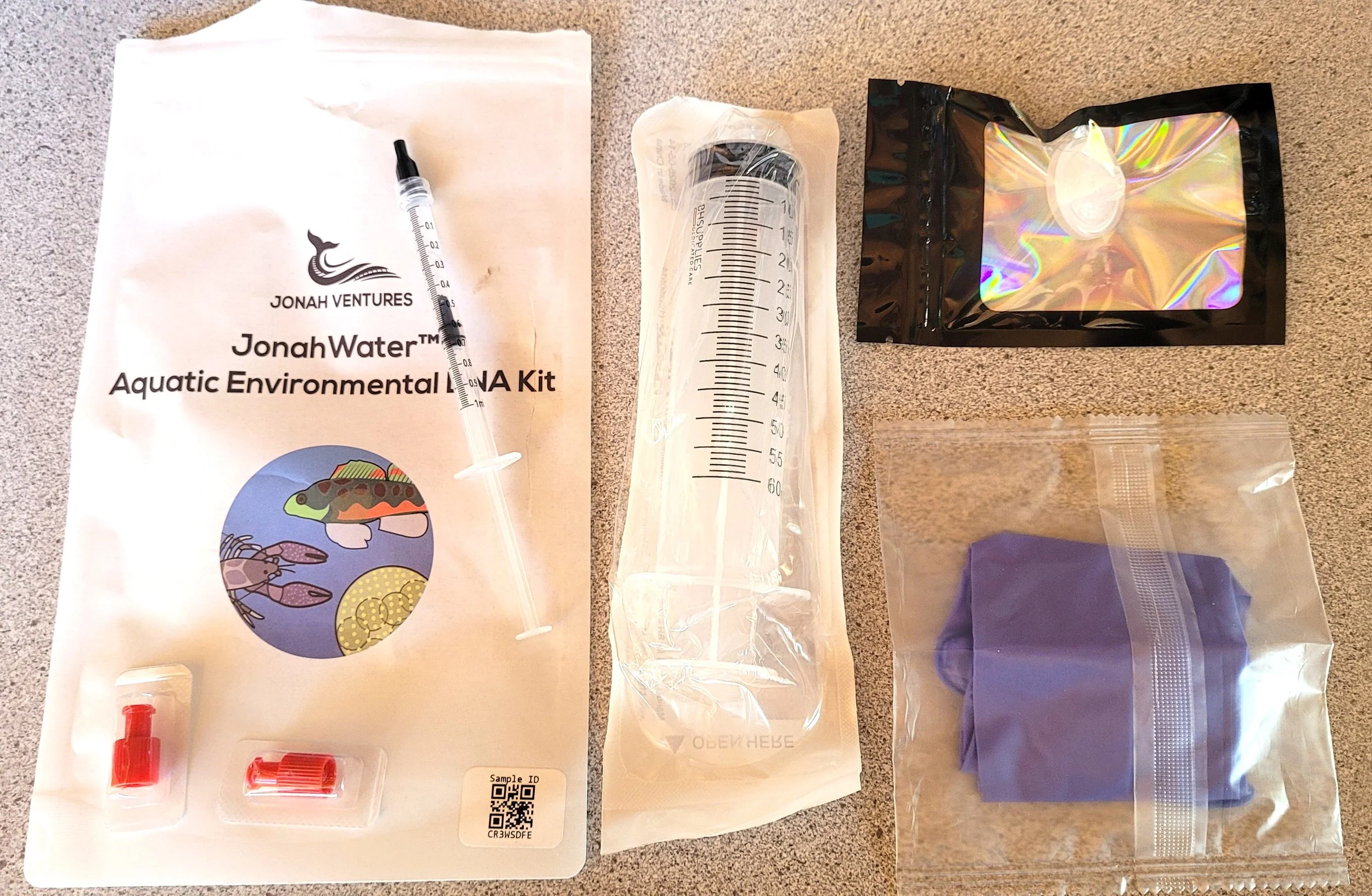
NFC prefers filters to water samples as they are less likely to get contaminated and are easier to handle and ship. See full sample test kit above
A syringe is used to force water through the filter until it is full. See top right below.
Sample Pull Plan Approval
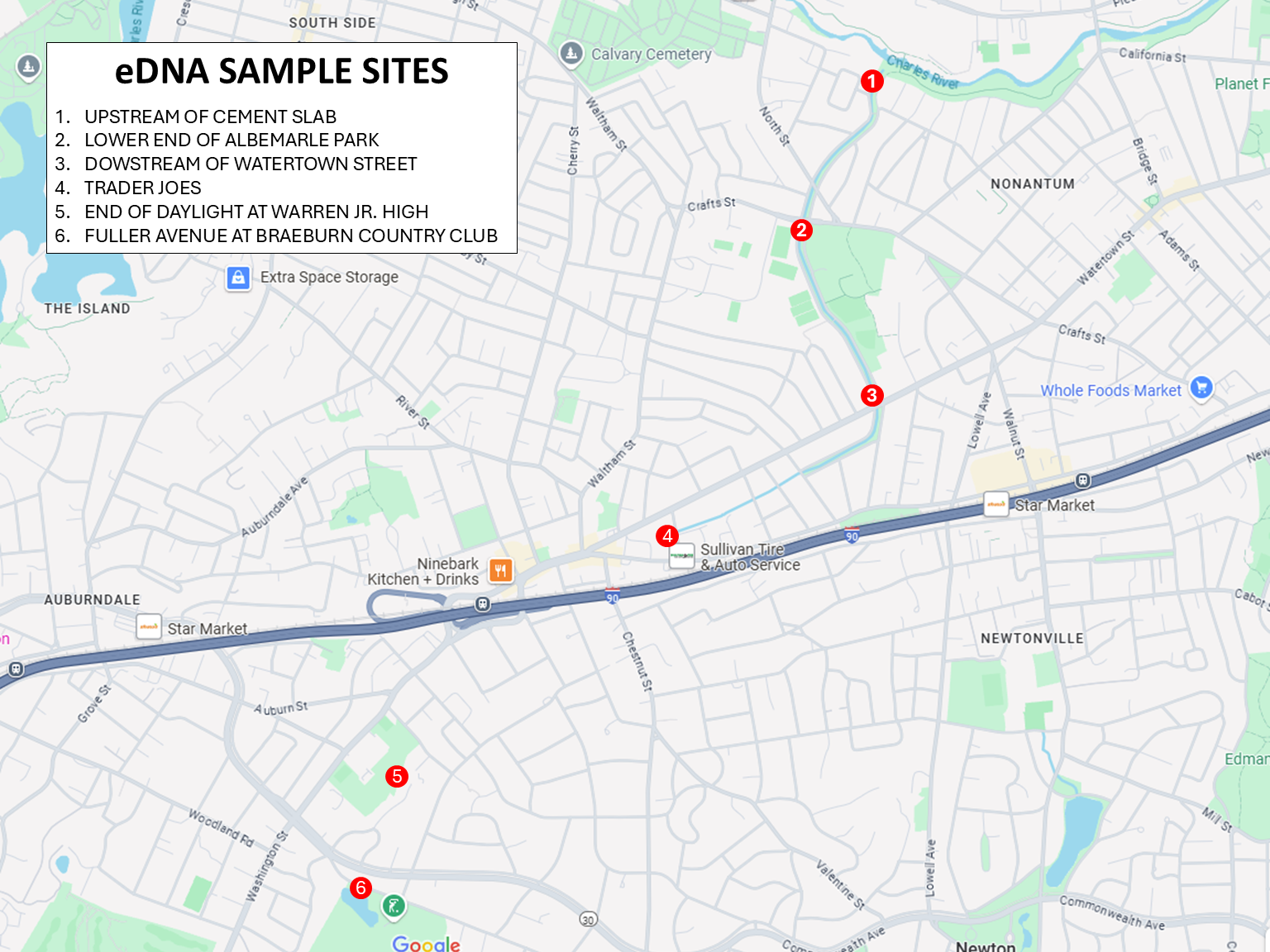
Proposed eDNA sample collection maps are required for approval and funding. We will require location names as they will appear on the return data, as well as GPS location.
All proposed sample sites should be visited prior to request submission to make sure that they are accessible.
If a site becomes inaccessible after submission and approval for any reason, we will require an updated collection site map before any samples are pulled.
Examples of Sample Maps:
eDNA Sample Submission
All eDNA samples must be provided to the appropriate lab for processing. It is important that samples be packaged per the lab specs, tracked, and confirmed when delivered.
eDNA samples should be hand delivered where possible.
This saves money and lessens the risk that the samples will be lost or damaged in transit.
All eDNA samples should be shipped USPS with tracking.
Jonah Ventures provides self-addressed envelopes for shipping samples.
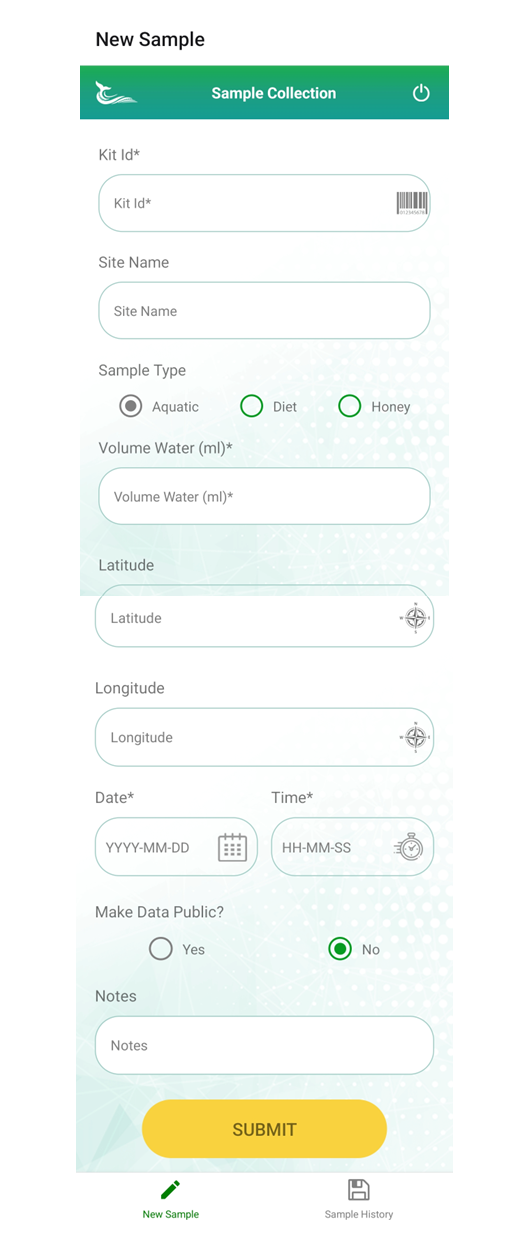
Some eDNA labs offer an app to upload information associated with samples such as site location name.
Submission apps should be used where available.
The screen scrape below comes from the Jonah Ventures app.
General Guidelines
Below are some general guidelines in regard to working with eDNA.
All related eDNA water samples should be pulled on the same day, and ideally within as short a time period possible. This is so that the location of positive hits are not impacted by fish movement.
When sampling streams, samples should be pulled from where the strongest current is.
The center if the stream is best in straight sections.
The outside edge is best when in meandering sections.
For larger streams and rivers, pull your samples from flowing areas while avoiding eddies and slow pools.
When targeting species known to inhabit pool in streams and rivers, pull your samples from immediately downstream of the pool.
When sampling small ponds and impoundments it is best to pull your sample from the outlet.
Always work downstream to upstream so as not to disturb the water.
Always pull your water sample from upstream of where you are standing.
Reporting
Most eDNA results are scientific in nature. The data is not typically conducive for disseminating to teh general public.
Most targeted eDNA results are presented in general text format.
Request tables where possible.
Excel is the preferred format for tables as most people have it.
Jonah Ventures eDNA metabarcoding data is presented in a series of tables. While they use Excel spreadsheets, the data is not in typical Excel format.
Create a new empty spreadsheet.
Copy and paste the Jonah data into the spreadsheet.
This should convert the data to standard Excel format.
Jonah Ventures eDNA metabarcoding data uses Latin names for species identification. Always provide common names for the general public.
Add a column for common species name and translate the Latin to English using the www as a source.
The result could be at the genus or species level.
Try to resolve reference data issues where the species data is obviously wrong.
For example, Jonah Ventures appears to report common blue jay as Florida jay.
Jonah Ventures eDNA metabarcoding data uses codes to represent sample site location. Always provide common names for the general public.
Example of Single-Location Modified Results:
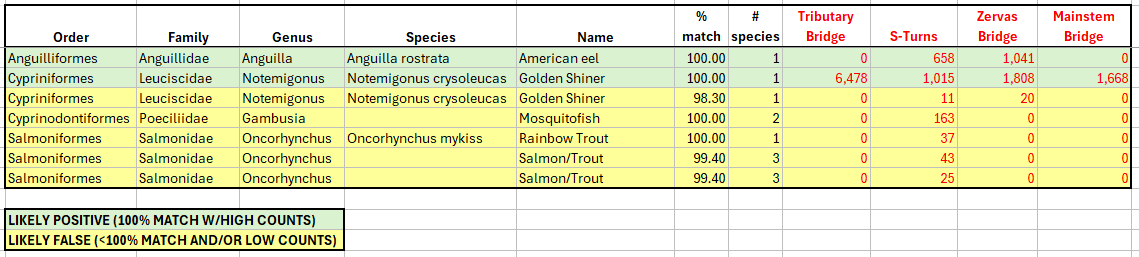
Example of Multi-Location Modified Results:
Summary
While a valuable tool, eDNA is both costly and complicated. If you are not familiar with its nuances, please reach out to National NFC for advise and direction. All eDNA projects must be pre-approved by National NFC as well.